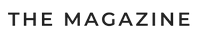The rise in price of energy resources stimulates the search for more efficient and cheaper types of fuel, including at the household level. Most enthusiastic craftsmen are attracted by hydrogen, whose calorific value is three times higher than methane (38.8 kW versus 13.8 with 1 kg of substance). The method of extraction at home, it would seem, is known - the splitting of water by electrolysis. In fact, the problem is much more complicated. Our article has 2 goals:
- to analyze the question of how to make a hydrogen generator with minimal costs;
- consider using a hydrogen generator for heating a private house, refueling a car, and as a welding machine.
Brief theoretical part
Hydrogen, aka hydrogen, - the first element of the periodic table - is the lightest gaseous substance with high chemical activity. During oxidation (that is, combustion) it emits a huge amount of heat, forming ordinary water. We characterize the properties of the element, designing them in the form of theses:
- Hydrogen burning is an environmentally friendly process, no harmful substances are released.

- Due to its chemical activity, free gas does not occur on Earth. But in the composition of water its reserves are inexhaustible.
- The element is extracted in industrial production by a chemical method, for example, in the process of gasification (pyrolysis) of coal. Often a byproduct.
- Another way to obtain gaseous hydrogen is the electrolysis of water in the presence of catalysts - platinum and other expensive alloys.
- A simple mixture of gases hydrogen + oxygen (oxygen) explodes from the slightest spark, instantly releasing a large amount of energy.
For reference. Scientists who first separated the water molecule into hydrogen and oxygen called the mixture explosive gas because of its tendency to explode. Subsequently, it received the name Brown's gas (by the name of the inventor) and became designated by the hypothetical formula of NGOs.

From the foregoing, the following conclusion suggests itself: 2 hydrogen atoms easily combine with 1 oxygen atom, but they are very reluctant to part. The chemical oxidation reaction proceeds with the direct release of thermal energy in accordance with the formula:
2H2 + O2 → 2H2O + Q (energy)
Here lies an important point that will be useful to us in the further analysis of flights: hydrogen enters into reaction spontaneously from ignition, and heat is released directly. To separate a water molecule, energy will have to be spent:
2H2O → 2H2 + O2 - Q
This is an electrolytic reaction formula that characterizes the process of splitting water by supplying electricity. How to put this into practice and make a hydrogen generator with your own hands, we will consider further.
Prototype creation
To make you understand what you are dealing with, first we suggest assembling the simplest hydrogen generator at the lowest cost. The design of a home-made installation is shown in the diagram.

What does a primitive electrolyzer consist of:
- reactor - glass or plastic container with thick walls;
- metal electrodes immersed in a reactor with water and connected to a power source;
- the second tank acts as a water shutter;
- HHO gas exhaust pipes.
An important point. The electrolytic hydrogen plant operates only on direct current. Therefore, use the AC adapter, car charger, or battery as the power source. An alternator will not work.
The principle of operation of the electrolyzer is as follows:
- Two electrodes immersed in water are supplied with voltage, preferably from an adjustable source.To improve the reaction, a little alkali or acid is added to the container (at home - ordinary salt).
- As a result of the electrolysis reaction, hydrogen will be released from the side of the cathode connected to the “minus” terminal, and oxygen will be generated near the anode.
- Mixing, both gases through a tube enter a water trap that performs 2 functions: separating water vapor and preventing a flash in the reactor.
- From the second tank, explosive gas of NNO is fed to the burner, where it is burned to form water.
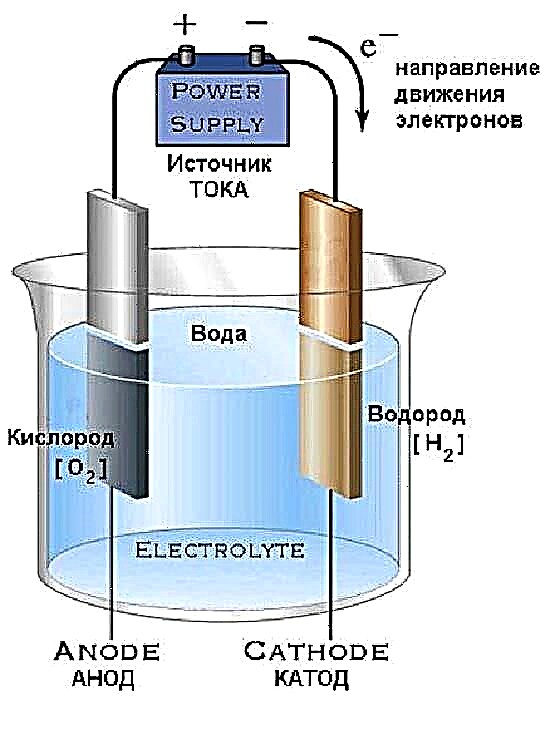
To make the generator design shown in the diagram with your own hands, you will need 2 glass bottles with wide necks and caps, a medical dropper and 2 dozen screws. A full set of materials is shown in the photo.

Of the special tools, a glue gun is required to seal the plastic covers. The manufacturing procedure is simple:
- Twist flat wooden sticks with self-tapping screws, placing their ends in different directions. Solder the screw heads together and connect the wires - get the future electrodes.
- Make a hole in the cover, insert the cut case of the dropper and wires there, then seal on both sides with a glue gun.
- Place the electrodes in the bottle and screw on the cap.
- In the second cap, drill 2 holes, insert the dropper tubes and screw onto a bottle filled with plain water.
To start the hydrogen generator, pour salted water into the reactor and turn on the power source. The onset of the reaction is marked by the appearance of gas bubbles in both containers. Adjust the voltage to the optimum value and set fire to Brown gas exiting the dropper needle.
About the Meyer hydrogen cell
If you made and tested the design described above, then by burning the flame at the end of the needle you probably noticed that the installation performance is extremely low. To get more explosive gas, you need to make a more serious device, called the Stanley Meyer cell in honor of the inventor.
The principle of operation of the cell is also based on electrolysis, only the anode and cathode are made in the form of tubes inserted one into another. The voltage is supplied from the pulse generator through two resonant coils, which allows to reduce the current consumption and increase the productivity of the hydrogen generator. The electronic circuit of the device is shown in the figure:

Note. Details on the operation of the scheme are described on the resource http://www.meanders.ru/meiers8.shtml.
To make a Meyer cell, you will need:
- a cylindrical case made of plastic or plexiglass; craftsmen often use a water filter with a lid and nozzles;
- stainless steel tubes with a diameter of 15 and 20 mm, length 97 mm;
- wires, insulators.

Corrosion-proof tubes are attached to the base of the dielectric, wires connected to the generator are soldered to them. The cell consists of 9 or 11 tubes placed in a plastic or plexiglass case, as shown in the photo.

The elements are connected according to all the schemes known on the Internet, which include an electronic unit, a Meyer cell and a water lock (technical name is the bubbler). For safety reasons, the system is equipped with sensors for critical pressure and water level. According to the reviews of home craftsmen, such a hydrogen installation consumes a current of the order of 1 ampere at a voltage of 12 V and has sufficient performance, although exact numbers are not available.

Plate Reactor
A high-performance hydrogen generator capable of ensuring the operation of a gas burner is made of stainless plates measuring 15 x 10 cm, the number is from 30 to 70 pcs. Holes for tightening studs are drilled in them, and a terminal for connecting the wire is cut out in the corner.

In addition to sheet metal grade 316, you will need to buy:
- 4 mm thick rubber resistant to alkali;
- end plates made of plexiglass or textolite;
- tie rods M10-14;
- check valve for gas welding apparatus;
- water filter under a hydrolock;
- connecting pipes from corrugated stainless steel;
- potassium hydroxide in powder form.

The plates must be assembled into a single unit, insulated from each other with rubber gaskets with a middle cut out, as shown in the drawing. Pull the resulting reactor tightly with studs and connect to the electrolyte nozzles. The latter comes from a separate container equipped with a lid and shutoff valves.
Note. We tell how to make a flow (dry) type electrolyzer. It is easier to make a reactor with immersion plates - no rubber gaskets are required, and the assembled unit is lowered into a sealed container with electrolyte.

Subsequent assembly of a hydrogen-producing generator is performed in the same way, but with differences:
- On the casing of the apparatus is attached a tank for the preparation of electrolyte. The latter is a 7-15% solution of potassium hydroxide in water.
- Instead of water, a so-called deoxidizer - acetone or an inorganic solvent - is poured into the “bubbler”.
- A check valve must be placed in front of the burner, otherwise, when the hydrogen burner is turned off smoothly, a reverse blow will break the hoses and the bubbler.
Is it profitable to get hydrogen at home
The answer to this question depends on the scope of the oxygen-hydrogen mixture. All drawings and diagrams published by various online resources are designed to produce HHO gas for the following purposes:
- use hydrogen as a fuel for cars;
- smokelessly burn hydrogen in heating boilers and stoves;
- apply for gas welding.
The main problem that crosses out all the advantages of hydrogen fuel: the cost of electricity for the release of pure matter exceeds the amount of energy received from its combustion. Whatever the adherents of utopian theories claim, the maximum efficiency of the electrolyzer reaches 50%. This means that 1 kW of heat consumed 2 kW of electricity. The benefit is zero, even negative.
 Recall that we wrote in the first section. Hydrogen is a very active element and reacts with oxygen on its own, generating a lot of heat. Trying to separate a stable water molecule, we cannot bring energy directly to atoms. The splitting is carried out by electricity, half of which is dissipated by heating the electrodes, water, transformer windings and so on.
Recall that we wrote in the first section. Hydrogen is a very active element and reacts with oxygen on its own, generating a lot of heat. Trying to separate a stable water molecule, we cannot bring energy directly to atoms. The splitting is carried out by electricity, half of which is dissipated by heating the electrodes, water, transformer windings and so on.
Important background information. The specific heat of hydrogen combustion is three times higher than that of methane, but - by mass. If we compare them by volume, then when burning 1 m³ of hydrogen, only 3.6 kW of thermal energy will be released compared to 11 kW for methane. After all, hydrogen is the lightest chemical element.
Now consider the explosive gas obtained by electrolysis in a homemade hydrogen generator as fuel for the above needs:
- The final installation price, low productivity and efficiency makes it extremely unprofitable to burn hydrogen for heating a private house. It is easier to put any of the electric boilers — TEN, induction or electrode — than to “wind” the meter with an electrolyzer.
- To replace 1 liter of gasoline for a car, 4,766 liters of pure hydrogen or 7,150 liters of detonating gas will be required, one third of which is oxygen. The most lousy inventor on the Internet has not yet made an electrolyzer capable of providing similar performance.

- A gas welding apparatus that burns hydrogen is more compact and lighter than cylinders with acetylene, propane and oxygen. Plus, the flame temperature up to 3000 ° C allows you to work with any metals, the cost of obtaining fuel here does not play a special role.
For reference. To burn hydrogen in a boiler, you will have to thoroughly redesign the structure, since a hydrogen burner can melt any steel.
Conclusion
The hydrogen in the composition of the NGO gas obtained from a home-made hydrogen generator is useful for two purposes: experiments and gas welding.Even if we discard the low efficiency of the electrolyzer and the costs of assembling it together with the consumed electricity, there is simply not enough productivity to heat the building. This also applies to the gasoline engine of a passenger car.